Thank you for downloading!
Subscribe to our newsletter to receive email updates about our work, get connected and support Australians impacted by eczema. You can unsubscribe at any time.
Subscribe NowIt’s only been a few weeks since we launched our SOS for Kids with Eczema campaign, and we already have important news to share.
We can confirm that the Pharmaceutical Benefits Advisory Committee (PBAC) will soon consider whether dupilumab (brand name Dupixent®) should be funded through the Pharmaceutical Benefits Scheme (PBS) for Australian children aged six months to 11 years with severe eczema.
As a health professional involved in the care of children with severe eczema, your voice can help decision-makers understand why timely access to effective treatment matters.

The PBAC welcomes input from healthcare professionals involved in the care of patients who may benefit from the medicine under review.
Health professionals see first hand the burden of severe eczema on children and families from the physical and emotional impact to the broader healthcare implications, including repeat doctor visits, referrals and frequent hospital admissions.
Your clinical experience and insights on the burden of severe eczema, the limitations of current treatment options and the importance of effective early intervention can help reinforce that severe eczema is not “just a rash” and that delays in access have real consequences for children and families.
Make a submission to the PBAC
You can provide input through the PBAC online questionnaire. Our healthcare professional toolkit includes guidance on how to do this and key points you may wish to highlight. Submissions close on 20 May.
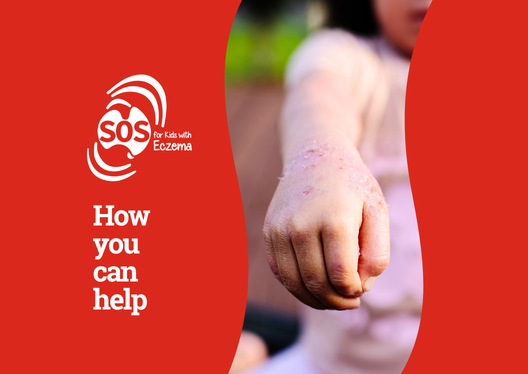
Together, we can help end the delay and inequity that Australian children with severe eczema have faced for far too long.

Subscribe to our newsletter to receive email updates about our work, get connected and support Australians impacted by eczema. You can unsubscribe at any time.
Subscribe Now