Thank you for downloading!
Subscribe to our newsletter to receive email updates about our work, get connected and support Australians impacted by eczema. You can unsubscribe at any time.
Subscribe NowThere is currently no cure for eczema. You may have heard that your eczema or your child’s eczema will go away as you get older, but this does not happen for everyone.
The good news is that there is lots you can do to manage eczema. It does not have to stop you or your child from enjoying life and doing the things you all want to do.
There are two treatments that work well together to manage eczema.
Both treatments are needed. This is because they help control eczema in different ways. When these two treatments are used well, they will control most eczema. To help plan your eczema management treatments download the Eczema Care Plan.
Sometimes other treatments are needed and we go into more detail elsewhere on our website and in our Eczema Care Online Toolkit.
These creams are used to get control of eczema. They are:
These creams are usually topical corticosteroid creams or ointments (often called steroid creams). Sometimes people are prescribed other flare control creams.
Most people with eczema will need to use flare control creams at some point.
If flare control creams are recommended by your doctor, nurse or pharmacist;
Are they safe?
Yes. Studies show that flare control creams are safe when used following the instructions above. They should be applied to the affected skin only.
For more information see: Topical Corticosteroids for Eczema Flare Control
When you have eczema, your skin loses moisture easily. Dry skin can lead to a new flare, infection or make existing eczema harder to manage. Moisturising every day helps protect and heal the skin barrier.
You may need to try a few different moisturisers to find the one that works best on your skin.
Avoiding triggers and looking after your mental health well-being is also important.
Here are some more eczema treatments your health professional may recommend:
For more information, ‘how to’ videos see our Helpful Links.
Here are some more eczema treatments your health professional may recommend:
For more information, ‘how to’ videos see our Helpful Links.
Treatment and Management
Medical researchers continue to strive for new treatments that provide relief from, what can be, an overwhelming experience. Be sure to ask your dermatologist about potential options and feel free to seek guidance from Eczema Support Australia. Eczema Support Australia is committed to helping people living with eczema overcome difficulties and thrive in their community.

Treatment and Management

Treatment and Management
Download Wet Dressings Information Sheet
Download Bleach Bathing Information Sheet

Treatment and Management
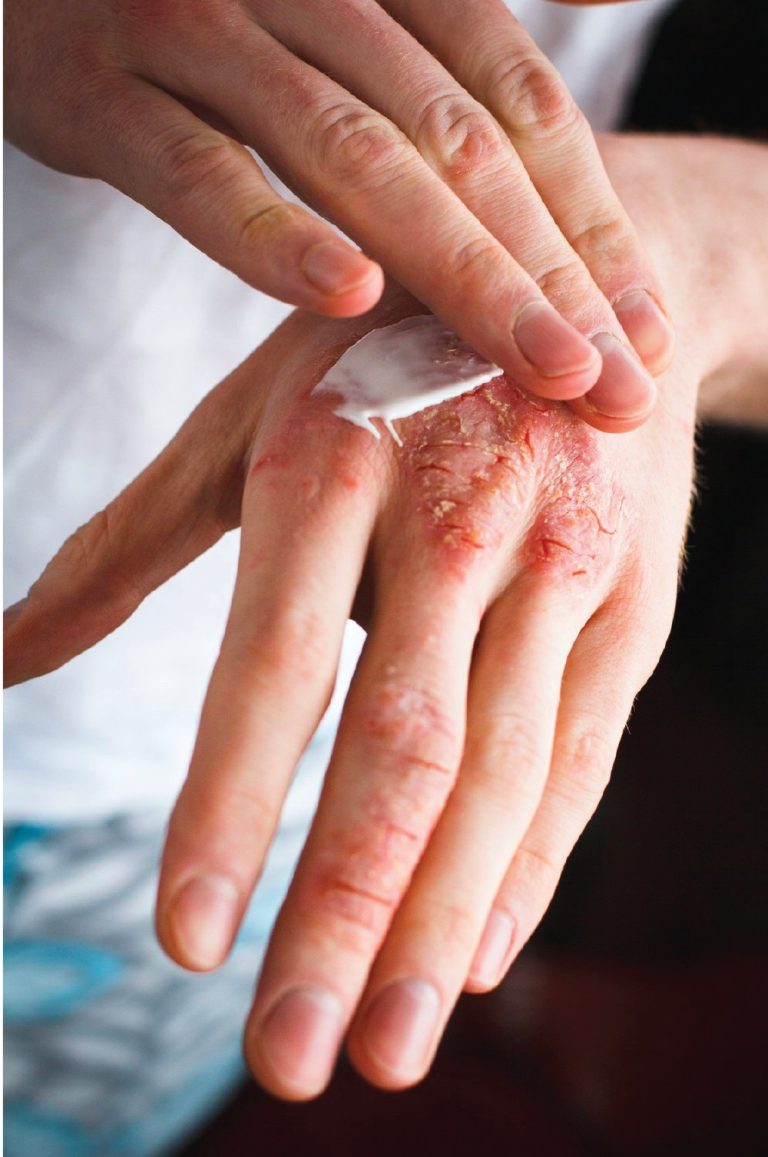
Hand eczema is an inflammatory condition that can cause itchy blisters, cracks, or rashes on the hands.
It can go hand-in-hand with atopic eczema or be a result of contact dermatitis.
Contact dermatitis can be more common in professions with frequent contact with water and chemicals. E.g. Hairdressers, cleaners, healthcare workers and hospitality workers.
Women are more likely than men to have another form of hand eczema called pompholyx or dyshidrotic eczema. This results in very itchy little blisters on the palms or fingers.

Treatment and Management
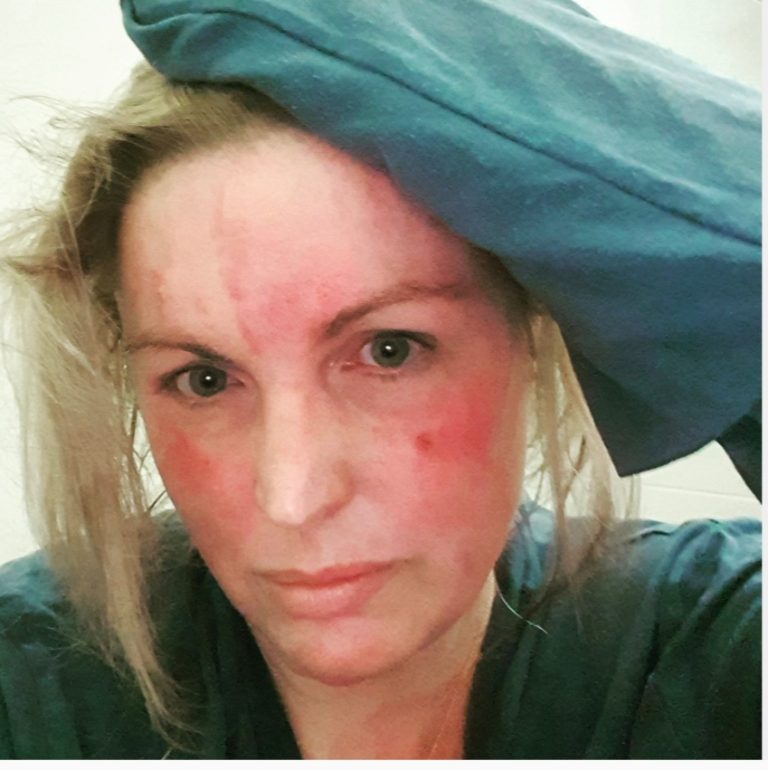
Facial eczema can be chronic, reoccurring, or a fleeting occurrence.
When eczema affects the face, it can have a huge impact on quality of life due to the sensitivity of this area of skin and the visibility.
In babies, cheeks and the folds of the neck are often affected by eczema. Children can develop further facial eczema, including the eyelids. Adults with facial eczema may have developed contact dermatitis.
Facial eczema is not always because of atopic or contact dermatitis. Facial eczema in adults may be a different form of eczema – seborrheic dermatitis. This can result in dry scaly skin around ears, eyebrows, eyelids or ear canals. It can also be the cause of scalp dandruff.
Eczema is a chronic and complex itchy skin condition. But it is more than just a skin condition. For those living with eczema, it can vary from a mild to moderate itch to an extremely itchy, painful and debilitating condition, with huge effects on quality of life. There are different types of eczema, the most common being atopic eczema.
Visit the Eczema Resource Library to learn more.
Use our Eczema Care Online Toolkit to find out the best way to care for your eczema or your child's eczema.
Subscribe to our newsletter to receive email updates about our work, get connected and support Australians impacted by eczema. You can unsubscribe at any time.
Subscribe Now